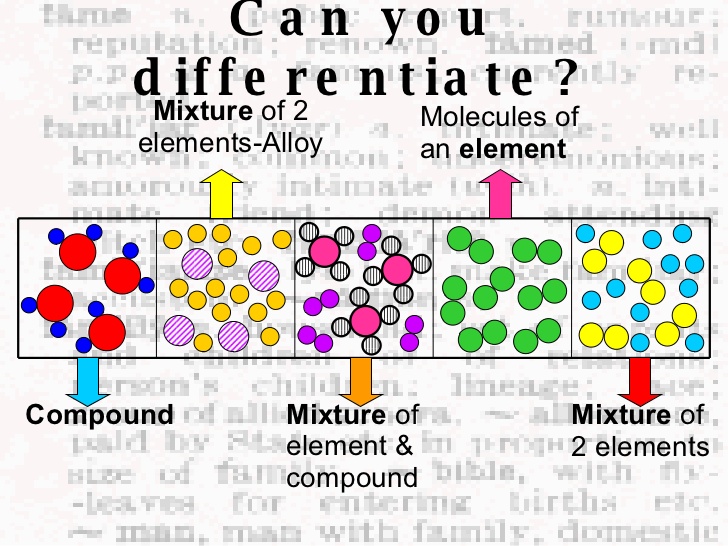 
An introduction of elements, compounds, and mixtures is crucial because it is a part of chemistry fundamentals. ConclusionĬhemistry is the study of how matter behaves and the factors that influence this behaviour. As the syllabus progresses, the concepts learnt in this introduction will be helpful in understanding more advanced concepts. This was an introduction of elements, compounds, and mixtures. Examples include mayonnaise, air, saline solution, most alloys, etc. Homogenous mixture: In these mixtures, the components cannot be distinguished and require special procedures to separate them. Examples include conglomerate rock, sand and iron filing powder, sand and water, a salad, etc. Heterogeneous mixtures: When the substances combined in the mixture can be easily seen and separated, the mixture is heterogeneous. Mixtures can be categorised into the following: Alloys, suspensions, colloids, and solutions are types of mixtures. In compounds, the electrons realign to form a substance with its properties, often different from the parent components. This means that the components retain their natural and chemical properties. When two or more substances are mixed but are combined by a chemical process, the result is a mixture. Intermetallic compounds that have metallic bonding.Ĭomplexes that have coordinate covalent bonds. Molecular compounds that have covalent bonds. These atoms are held together by chemical bonds based on which compounds can be classified as: These are important because they also form salts necessary for the survival of organisms.Ĭhemical compounds are made up of atoms that combine in a fixed ratio, and the molecules thus formed occupy a defined space. 
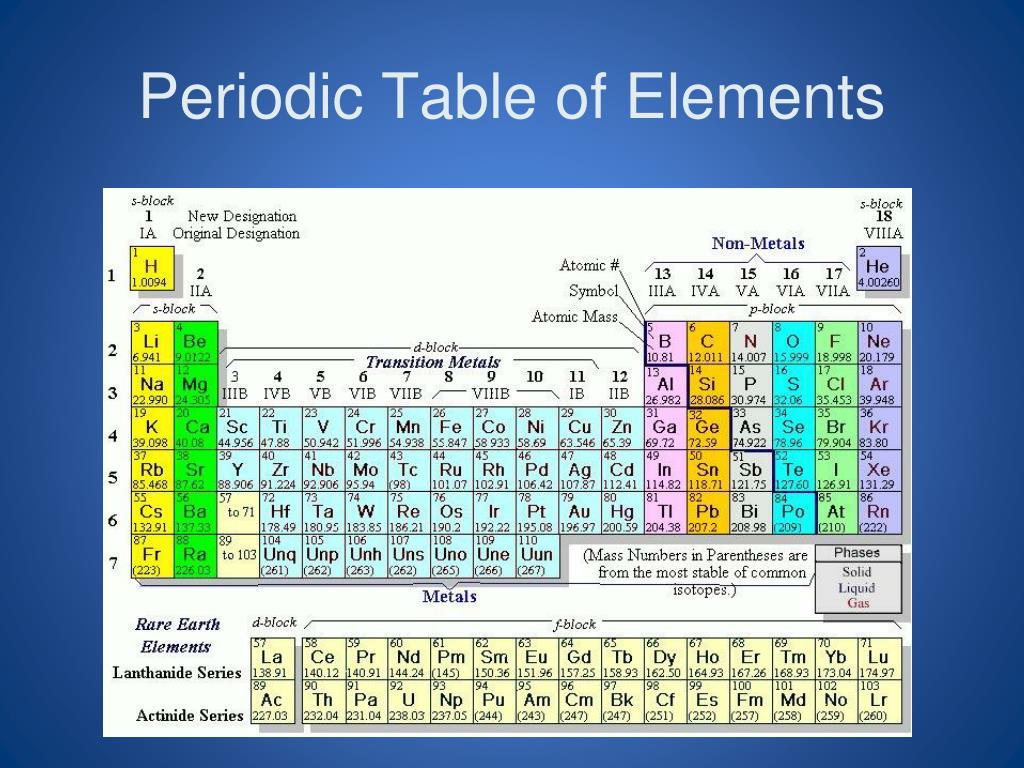
These compounds play an essential part in the life of our planet since it is through a combination of elements and compounds that the building blocks of all organic matter are formed. Most elements on earth combine to form compounds. This process is known as chemical bonding. When elements combine through chemical processes, bonds are formed between their atoms by a sharing or transference of electrons. Even so, the most agreed upon metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium.Ī chemical compound results from two or more elements combining through a chemical process. There is no universally recognised definition of metalloids. Metalloids: These are elements that have properties that are a mixture of both metals and non-metals. Some examples of non-metals are Fluorine (F), Chlorine (Cl), Iodine (I), etc. They share specific properties that are used to categorise them as non-metal. Non-metals: These elements are placed on the right side of the periodic table. Some examples of metals are Iron (Fe), aluminium (Al), copper (Cu), mercury (Hg), and lead (Pb). They are typically ductile or can be drawn into wires.Īre malleable or can be beaten into sheets. These elements share some characteristics such as: Metals: Metallic elements are usually found on the left side of the periodic table. Elements are placed in the periodic table according to their atomic number.Įlements can be categorised into metals, non-metal, and metalloids in the periodic table. This number of protons represents the atomic number of the element. All the atoms of an element will have the same number of protons in their nucleus. In chemistry, an element is a pure substance made up of only one type of atom. It is essential to introduce elements, compounds, and mixtures since these form the basis of studying matter and its properties. The components of a mixture do not form chemical bonds like they do in a compound. They are substances formed by physically mixing other substances. Pure water extracted from a plant and obtained by purifying tap water will be made up of a combination of hydrogen and oxygen that is constant irrespective of the source. Elements and compounds have a constant chemical composition, and thus they have uniform chemical properties. Pure substances can be further broken down into elements and compounds. 
All the matter in our knowledge can be broken down into pure substances and mixtures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
